It may not happen automatically, because it might have a large activation energy that slows it down or stops it from taking place at a certain temperature. Essentially, a feasible or spontaneous reaction is one that is energetically possible. The more ways there are, the higher the amount of entropy.Įarlier we said that for a reaction to be feasible, energy must move in the direction of increasing total entropy. So when we talk about entropy we are talking about the amount of possible ways energy can be distributed among the molecules in a system.

As you have seen from the example of the gas particles, this energy is distributed randomly. If you were to increase the amount of total energy in the system, for example by heating a gas, you would increase the number of available quanta that a particle can have.Įntropy, Olive StudySmarter OriginalsĮssentially, you can say that the more energy a system has, the more ways there are to distribute energy between the molecules. The amount of quanta available to a particle in a system is limited to the total energy of the system. In other words, every time you take a snapshot, there is a new arrangement of quanta between the molecules. You must imagine that as the particles interact with each other, the quanta get distributed (or spread out) among them. A particle can only have a whole number of quanta, never fractions of quanta. Įnergy exists in ‘packets’ which we call quanta. Gas particles constantly interact and transfer energy with each other. As a result, one molecule could have a certain amount of energy in one snapshot but have less in another one. The faster particles have more energy than the slower ones. Each collision causes some to speed up while others slow down. Think of how gas particles constantly collide with each other and the walls of a container. This is because the molecules constantly interact and transfer energy with one another. However, if you could take snapshots of the molecules in a system at given instances in time, you would find that the molecules rarely have the exact same energy. To help you understand entropy, consider that molecules at equilibrium have the same average energy. Calculate the entropy change of a given reaction using standard entropy values.Discover how the second law of thermodynamics helps to explain spontaneous reactions.You will also learn about spontaneous or feasible processes.Why do we say entropy is about energy distribution between particles?.You will learn the definition of entropy.Reaction Quotient and Le Chatelier's PrincipleĮntropy (S) is the number of possible ways energy can be distributed in a system of molecules.Elemental Composition of Pure Substances.Application of Le Chatelier's Principle.
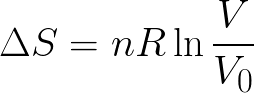
Intramolecular Force and Potential Energy.Variable Oxidation State of Transition Elements.

